•Prevention of "Glass Clouding"
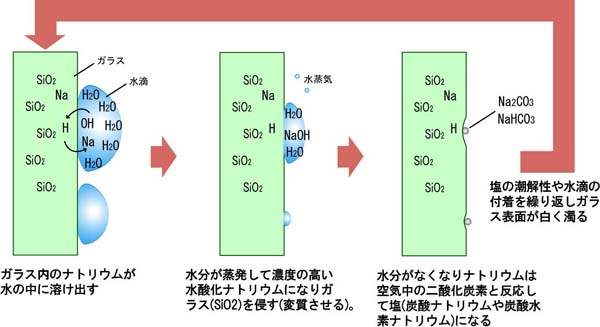
Since glass clouding occurs due to moisture, it can be improved by not allowing moisture to attach to glass surfaces, or wiping immediately if attached, minimizing Na dissolution, or keeping in constant contact with large amounts of water, but this is not realistic depending on use and installation environment. It can also be improved with coatings, etc., but coating maintenance itself becomes necessary and is not perfect. In chemical experiments, since components dissolving from glass may interfere with experiments, and heat resistance against heating and heat generation is required, glass containers with different components from window glass (soda-lime glass), such as borosilicate glass and quartz glass, are used. Using such glass for windows would also improve clouding, but it becomes very expensive. Understanding that glass also weathers depending on environment, we recommend cleaning as much as possible within reasonable limits and washing away attached salt.